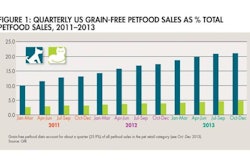
HACCP CHECKLIST
1. Prerequisite Programs
1.1 Are all PRPs adequately defined and documented (a GMP
standard should be used to verify this)?
1.2 Are PRPs regularly monitored and verified?
1.3 Have PRPs been validated where applicable (e.g. cleaning
methods and schedules, hand sanitizing, raw material
inspection, etc.)?
1.4 Has complete traceability been established?
1.5 Is effective vendor auditing regularly performed?
1.6 Is adequate training performed and is its effectiveness
monitored?
2. HACCP Team
2.1 Has a qualified food safety team leader been appointed?
2.2 Has the team received adequate training?
2.3 Does the team have enough scientific expertise to address
all hazards (biological, chemical, physical)? If not, have
suitable consultants been employed?
2.4 Does the team meet at regular intervals? Are minutes kept?
Are team decisions carried out in a timely manner?
3. Description of Product
3.1 Have all ingredients and sub-ingredients been identified?
3.2 Have microbiological and chemical characteristics been
established and are they validated?
3.3 Has the intended use been properly defined?
3.4 Has the possibility of inadvertent misuse been
identified?
4. Flowcharts
4.1 Do flowcharts cover the entire process including
sub-contracting, external warehousing, rework, special
products, limited editions, etc.?
4.2 Are raw ingredients and packaging materials included in the
flowchart?
4.3 Are flowcharts detailed enough to allow adequate hazard
identification?
4.4 Are flowcharts verified?
5. Hazard Identification
5.1 Have all potential hazards been identified and documented?
5.2 Have hazards associated with ingredients been identified
and documented?
5.3 Have hazards associated with nutritional inadequacy been
identified and documented?
5.4 Are hazards specific (species of bacteria, type of
contaminant…)?
6. Hazard Evaluation
6.1 Has an adequate methodology for determining risk been
adopted? Is the methodology based on evaluation of at least
likelihood and severity?
6.2 Has the correct level of severity been assigned to each
hazard?
6.3 Has the correct level of likelihood been assigned to each
hazard? Has supporting evidence been provided?
6.4 Has significance been determined based on likelihood and
severity and is this determination compatible with the
methodology?
6.5 Has the acceptable level been determined for each
significant hazard? Has this been justified?
7. Identification of Control Measures
7.1 Has a methodology for determining CCPs, such as a decision
tree, been adopted? Is it adequate?
7.2 Has each significant hazard been assigned a CCP or an oPRP?
7.3 Has the effectiveness of the control measure applied at
each CCP or oPRP been scientifically validated? Are validation
records available?
7.4 Are all employees familiar with the CCPs and/or oPRPs?
8. Critical Limits
8.1 Have critical limits been set for each CCP?
8.2 Have the critical limits been scientifically validated? Are
validation records available?
9. Monitoring
9.1 Have monitoring practices been written in a procedure?
9.2 Is monitoring performed by skilled employees with a
thorough understanding of the CCP and its importance?
9.3 Is the actual control measure being monitored? The hazard
itself should not be the target for monitoring.
9.4 In batch production, does monitoring cover every single
batch?
9.5 In continuous processes, continuous on-line monitoring is
preferable. If this is not possible, is monitoring frequency
adequate? When periodic monitoring is performed, frequency must
be high enough ensure sufficient control in case deviation is
detected.
9.6 Are oPRPs adequately monitored?
9.7 Are monitoring activities properly recorded and are records
kept at least throughout the product's shelf life?
10. Corrective action
10.1 Are there clear and detailed written guidelines describing
exactly must be done in the event of a deviation for each CCP
and oPRP?
10.2 Are relevant employees familiar with these guidelines?
10.3 Have corrective action guidelines been validated to prove
their adequacy?
10.4 Do the corrective action guidelines describe what to do
with potentially non-conforming material? i.e. how it is
segregated, marked and blocked?
10.5 Do the guidelines describe in detail all of the possible
dispositions for non-conforming materials? i.e. rework,
reprocess, re-designate, destroy, etc.
10.6 Is it clear who has authority to decide on the destiny of
non-conforming material and what criteria are applied?
10.7 Do the guidelines provide guidance on how to resume
operation following a CCP deviation?
10.8 Do the guidelines require that action be taken to
eliminate root causes of failure in order to prevent
recurrence?
10.9 In organizations certified to ISO 9001, are the corrective
action guidelines compatible with clause 8.3 (control of
non-conforming product) and clause 8.5.2 (corrective action)?
10.10 Are corrective action events properly recorded and are
records kept at least throughout the product's shelf life?
11. Verification
11.1 Are written verification procedures in place for each PRP,
CCP and oPRP?
11.2 Do the verification activities provide adequate proof that
all elements of the food safety management system are actually
in place? Is there a process in place to prove that monitoring
is being performed as required?
11.3 Is there a pre-shipment release procedure in place,
providing assurance that each batch of finished product has
been adequately controlled at each CCP?
11.4 Is there a process in place to verify that measuring
devices used for each CCP are calibrated and accurate? If a
deviation in measuring equipment accuracy is detected, is
corrective action as described in item 10 above initiated?
11.5 Do the verification activities provide adequate proof that
the goals of the food safety management systems are being
achieved?
11.6 Are verification activities recorded and are records kept
at least throughout the product's shelf life?
11.7 Does verification include at least internal audits, trend
analysis, analysis of customer complaints and routine product
testing?
11.7 Is there evidence of corrective action when verification
shows inadequacy of any element of the food safety management
system?