
Copper, zinc and manganese are metal cations with numerous essential functions, but their predominant roles are as structural or catalytic components of enzymes. To that end, each one plays a role in both human and animal nutrition.
Copper, for example, is essential for energy production, connective tissue formation, antioxidant defense, pigment formation, iron transport, and hormone and neurotransmitter synthesis. Copper will also influence reproduction.
Zinc has structural and catalytic functions in proteins, including enzymes involved in bone synthesis, resorption and remodeling; the protein metallothionein, which plays a role in intracellular metal metabolism and storage (Cousins et al., 2006; Davis and Cousins, 2000); and as a modulator of immune function. Zinc also modulates the inflammatory response (Peterson et al., 2008) and affects the development of immune organs and functionality of immune cells (Rink and Haase, 2007). Zinc has a direct impact on the quality of the skin and fur coat of the animal.
Manganese provides antioxidant protection (de Rosa et al., 1980) and is a cofactor for many other enzymes (Au et al., 2008). Like zinc and copper, manganese has functions in bone metabolism (Beattie and Avenall, 1992).
Advancements in the science of nutrition are occurring seemingly monthly. One such advancement has been a development in the field of micronutrient absorption that is extremely important for companion animal diets. While there are several sources of trace elements approved for use in feeds, not all are equally “available” to animals. The best-known and most widely used trace minerals are inorganic sulphates and oxides. These vary greatly in their solubility or, in more explicit terms, their tendency to dissolve in the aqueous medium of the digestive tract.
Subsequently, sulphates are superior to oxides in this regard. Copper oxide would therefore be attributed a very low solubility score. In fact, being very tightly bound, it would be almost completely unavailable to an animal. While sulphates are comparatively quite soluble, they are also quite unstable in feed and break down over time, causing an increase in oxidation that can lead to spoilage and fat rancidity, as well as degradation of sensitive ingredients such as vitamins. This breakdown is associated with the formation of harmful free radicals and peroxides and reduced palatability of feed (Miles et al. 1998; Lu et al. 2010; Pang and Applegate 2006; Shurson et al. 2011).
Chelated (or complexed) minerals and hydroxy trace minerals are two categories representing relatively newer technologies of trace minerals that offer several advantages over traditional sulfate and oxide minerals. Chelated minerals are micronutrients that are paired with an organic ligand to enhance absorption. There are a number of potential ligands for mineral binding, including amino acids, peptides and polysaccharides, and each has a slightly different bio-availability. Chelated minerals are known as “organic minerals.”
Until recently, trace minerals have been categorized as either inorganic or organic; sulphates and oxides, or chelated and complex. Beginning in the mid-1990s, a new category of trace minerals known as hydroxy trace minerals received Association of American Feed Control Officials (AAFCO) approval (and, more recently, EU approval) as feed additives for all animal species, including dogs and cats. These products have similar solubility and availability profiles as the best organic trace elements.
They contribute to feed stability in that they are not soluble at neutral pH, making them more stable in the intestinal tract. This differentiation results in fewer reactive trace compounds that can promote oxidation or binding with other feed components, leading to optimized overall nutrient availability. The crystalline structure of hydroxy minerals provides slow release over the full length of the small intestine, resulting in more efficient trace element absorption and improved intestinal health.
While it can be difficult to directly compare various bioavailability trials due to variation in experimental design, it is generally accepted that hydroxy and high-quality chelated minerals improve micronutrient absorption by 10–20%, and even more when antagonists are plentiful. Another advantage to hydroxy and high-quality chelated minerals is their reduced toxicity compared to sulphated minerals. Both hydroxy and high-quality chelated minerals are more stable in feed, thereby reducing damaging free radicals that can lower the palatability of the feed (see Figure 1) while protecting other expensive nutrients in the feed (see Figure 2).
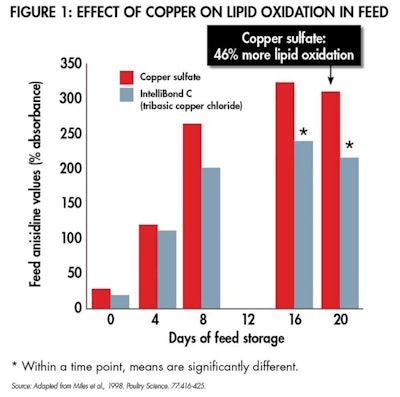
Both hydroxy and high-quality chelated minerals are more stable in feed, thereby reducing damaging free radicals that can lower palatability.
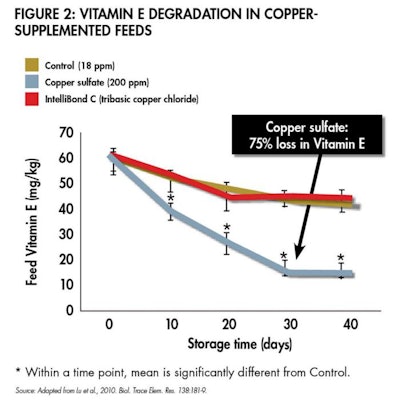
The same minerals can also reduce damage to other nutrients in feed, such as vitamins.
The supplementation of trace elements plays an essential role in maintaining optimal health and performance in all animals. As the type of trace element employed in the full feed can affect bio-availability, degradation of other feed ingredients, and shelf life, and can influence repeat sales due to owner satisfaction (or lack thereof), this decision can be considered both extremely important and highly marketable. Overall feed performance can definitely be improved by the use of high-quality trace mineral sources such as organic chelates or hydroxy trace elements.
Practically speaking, hydroxy trace minerals are nutritionally competitive with organic minerals, have AAFCO and European Union (EU) approval, and are significantly less expensive. They are worth serious consideration.
More on pet food ingredients


















