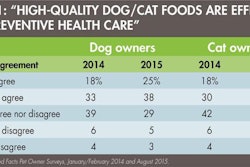
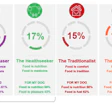
The final rule from the US Food and Drug Administration (FDA) for implementation of preventive controls for animal feed (including pet food) under the Food Safety Modernization Act (FSMA) was published in the Federal Register in September 2015. Compliance dates for some businesses on some aspects are as early as September 2016. Thus, it behooves the pet food manufacturer to become familiar with the new requirements and begin the process of incorporating these practices with due haste.
The Federal Register notice itself is over 660 pages in length. Much of that is comprised of FDA's response to comments to the proposed rule, and the actual new regulations don't start until the last 80 pages or so. Still, that's quite a read! Here's a very brief synopsis of some of the new requirements:
- cGMPs: FDA has established Current Good Manufacturing Practices (cGMPs) for all animal feed facilities. Before FSMA, only canned pet food operations had to comply with specific cGMPs. The new rules touch on virtually all aspects of pet food production, from storage of ingredients and finished product to proper maintenance of equipment to the need for adequate toilet facilities for employees, all with the focus on the prevention of contamination. Specific attention is given for the processing of by-products from the human food industry for use in animal feed. FDA understands the unique aspects of the animal feed industry as compared to the human food industry and appreciates the need for some degree of flexibility. While the new requirements may not be as onerous in all respects as those being put in place for human food production under the same law, it still will take a lot of adjustments to bring many companies into compliance.
-
Preventive controls: Companies must prepare and implement a written food safety plan. Integral to it all is proper documentation and recordkeeping. If it's not written down, it doesn't exist. Components of this aspect of FSMA include:
- Written hazard analyses
- Written preventive controls
- Written supply chain programs
- Written recall plans
- Written procedures for monitoring the implementation of the preventive controls
- Written corrective action procedures
- Written verification procedures
The timeframe for compliance with these new rules depends on the size of the company. A "small business" is defined as one with fewer than 500 full-time equivalent employees. A "very small business" averages less than US$2,500,000 in sales (plus the market value of food produced but held without sale) per year. Those pet food manufacturers who fit within one of these categories have more time to conform:
- Other than small or very small: one year for cGMPs, two years for preventive controls
- Small: two years for cGMPs, three years for preventive controls
- Very small: three years for cGMPs, four years for preventive controls (except for records to document "very small" status)
Compliance dates for receiving facilities are also affected by when the supplier is required to comply. For example, a small business may not have to held to the three-year allowance for implementation of the preventive controls if the supplier of that business is not required to comply by that date (i.e., it is "very small"). However, in that case controls must be put into place no more than six months after the supplier is required to comply.
FDA has developed or is developing guidance documents and training programs to assist pet food manufacturers in complying with these new rules. There have been many webinars and other training available through major trade associations and other non-governmental bodies, even before the new rules were finalized. There should be a wealth of information available to pet food manufacturers on this topic in the coming years.
The FDA Amendments Act (FDAAA) was enacted in 2007, well before FSMA. It required, among other things, that FDA promulgate regulations to address perceived deficiencies in ingredient, processing and labeling standards for pet foods. While some other aspects of FDAAA have been implemented for some time (e.g., the Reportable Food Registry), FDA has yet to even propose regulations to address these other issues. They were required by FDAAA to be implemented by 2009.
FDA has little control on some aspects of the rulemaking process, such as when any proposals are sent outside the agency for review. With the passage of FSMA in 2011, it was given the higher priority and any progress on FDAAA was sidelined. Still, an FDA official commented earlier in 2015 that the agency expected to propose new regulations as required by FDAAA by the time the final rule for FSMA was published. Well, that happened, yet to date nothing has been published. The pet food manufacturer has to wait longer to get a glimpse as to what other changes may be coming down the road.
Food Safety Modernization Act and Animal Feed
www.fda.gov/AnimalVeterinary/Products/AnimalFoodFeeds/ucm347941.htm